Contemporary Challenges: Fall-2023
Nuclear Physics Practice Problems (SOLUTION): Due 30 Wednesday
- Practice Nuclear Physics
S1 4749S
1. Rutherford scattering
Based on Q13M.2 from Unit Q 3rd Edition
Estimate the minimum kinetic energy that an alpha particle must have to get stuck to a gold nucleus? Give your answer in both joules and electron volts.(Note: The atomic number of gold is \(Z = 79\), the mass number of gold is \(A = 197\). An alpha particle consists of 2 protons and 2 neutrons. This question can be solved using knowledge of the Coulomb potential for a system of two point charges).
An alpha particle can get inside a gold nucleus if its initial kinetic energy is large enough to overcome the electrostatic repulsion between the positively charged gold nucleus and the positively charged alpha particle. We need to calculate the electrostatic potential energy when the surface of an alpha particle is about to touch the surface of a gold nucleus. The nuclear radius of gold is estimated from \(r_{\text{Au}}=r_{0} A^{1 / 3}=7.27\ \mathrm{fm}\) where \(A=197\) is the mass number and \(r_0 = 1.2 \text{ fm}\). The nuclear radius of the alpha particle is estimate by \(r_{\text{alpha}}=r_{0} 4^{1 / 3}=1.9\ \mathrm{fm}\). Therefore, the kinetic energy that the alpha particle needs is \begin{align} KE_{\alpha}&=k \cdot \frac{2 e \cdot 79 e}{ r_{\text{Au}}+r_{\text{alpha}}}\\ &=4 \times 10^{-12} \text{ J}\\ &=25\text{ MeV} \end{align}
where \(k\) is the Coulomb's law constant \(9 \times 10^9\) J.m/C\(^2\).
-
2. Fusion
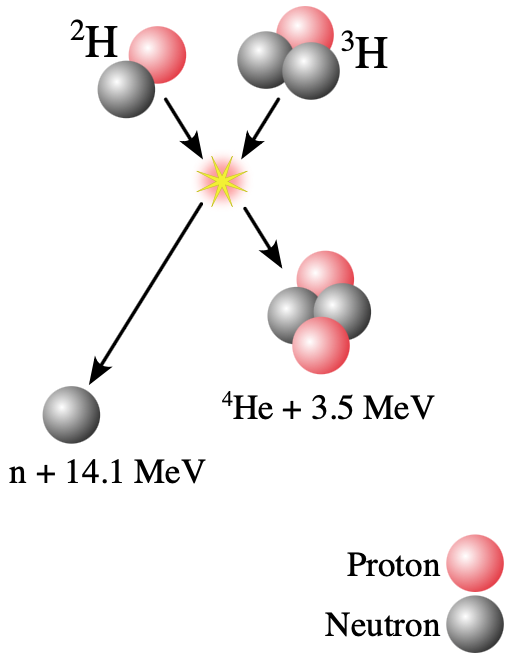
- The figure shows the fusion reaction between tritium and deuterium. Estimate the electrostatic potential energy of a deuterium-tritium pair at separation distance where the strong nuclear force becomes significant (i.e. the nuclei "touch" and the strong force can stick them together). Give a numerical answer in joules.
- Fusion is happening in a hot ionized gas of deuterium and tritium (also known as a D-T plasma). The average kinetic energy of the individual deuterium and tritium ions is about 10 times smaller than your answer to part a. (Fusion can be sustained by a small fraction of nuclei that have higher-than-average kinetic energy). Estimate the temperature of the plasma using the relationship:
Average kinetic energy of gas atom \(=(3/2)k_BT\),where \(k_B = 1.38 \times 10^{-23}\) J/K is called the Boltzmann constant. Give a numerical answer in kelvin.
- Compare the mass of \(^3\)He (a nucleus with two protons and one neutron) to the combined mass of two isolated protons and one isolated neutron. You are welcome to reference emperical data, but the main task of this question is to estimate the mass difference using the coarse-grained model we discussed in class. In this coarse-grained model, the biggest contributions to binding energy are the electrostatic interaction and the strong interaction.
- Estimate the mass difference and give your answer in units of kilograms.
- Is the mass of the composite particle larger or smaller than the mass of the individual parts?
- Using the approximation method for finding nuclear radius: \(r_1=1.51\) fm and \(r_2=1.72\) fm. \begin{align} U_{electric}&=k \cdot \frac{e^2}{ [3.23 \times 10^{-15} \text{ m}]}\\ &= 7.1 \times 10^{-14} \text{ J} \end{align} where \begin{align} k=9\times 10^9 \text{ J.m/C}^2\\ e=1.6\times 10^{-19} \text{ C}\\ \end{align}
From equipartition theorem (which we will study later), we expect each particle in a gas to have kinetic energy \begin{align} KE \approx (3/2)k_BT\\ \end{align} We are looking for a temperature where an average deuterium or tritium ion has \(KE \approx 7.1 \times 10^{-15} \text{ J}\). Therefore, \begin{align} T &= (2/3) \frac{[7.1 \times 10^{-15} \text{ J}]}{3k_B}\\ &= \frac{2 \cdot [7.1 \times 10^{-15} \text{ J}]}{3\cdot [1.38 \times 10^{-23} \text{ J/K}]}\\ &= 3.4\times 10^{8} \text{ K}\\ &= 340 \text{ million K} \end{align}
- Solution hasn't been writen yet.